Dec 12, 2024
In the future, climate-friendly hydrogen will replace fossil fuels in the mobility sector, in industry and in municipal energy projects around the world. In Germany alone, the production capacity of green hydrogen is to be increased to ten gigawatts by 2030. In order to accelerate the international hydrogen ramp-up and quickly build up sufficient production capacity for green hydrogen, it is important to use the right technology. We look at the advantages and disadvantages of alkaline electrolysis and PEM electrolysis.
Green hydrogen is the “fuel of the energy transition”. Unlike fossil fuels, no CO2 emissions are caused during its production and also later during its use. It is important that the energy for hydrogen production comes from renewable energy sources such as photovoltaic or wind power plants. Renewable energies, green hydrogen and its derivatives such as ammonia and methanol are particularly essential for the energy transition in industry and for heavy-duty transportation, such as trucks, shipping and aviation.
Electrolysis, the splitting of water into hydrogen and oxygen using electricity, has proven to be the most suitable process for the production of green hydrogen. There are various electrolysis processes: PEM electrolysis based on proton exchange membrane (PEM) technology, and alkaline electrolysis (AEL). Those are the most mature processes. Other electrolysis processes include high-temperature electrolysis and AEM water electrolysis (AEM stands for Anion Exchange Membrane).
While AEM and high-temperature electrolysis have not yet reached full technical maturity and are only operated in pilot projects or on a small scale, alkaline electrolysis and PEM electrolysis are particularly important for the rapid hydrogen ramp-up. PEM electrolysis in particular is one of the most important processes for industrial-scale hydrogen production from renewable energy sources.
What happens in the electrolyzer?
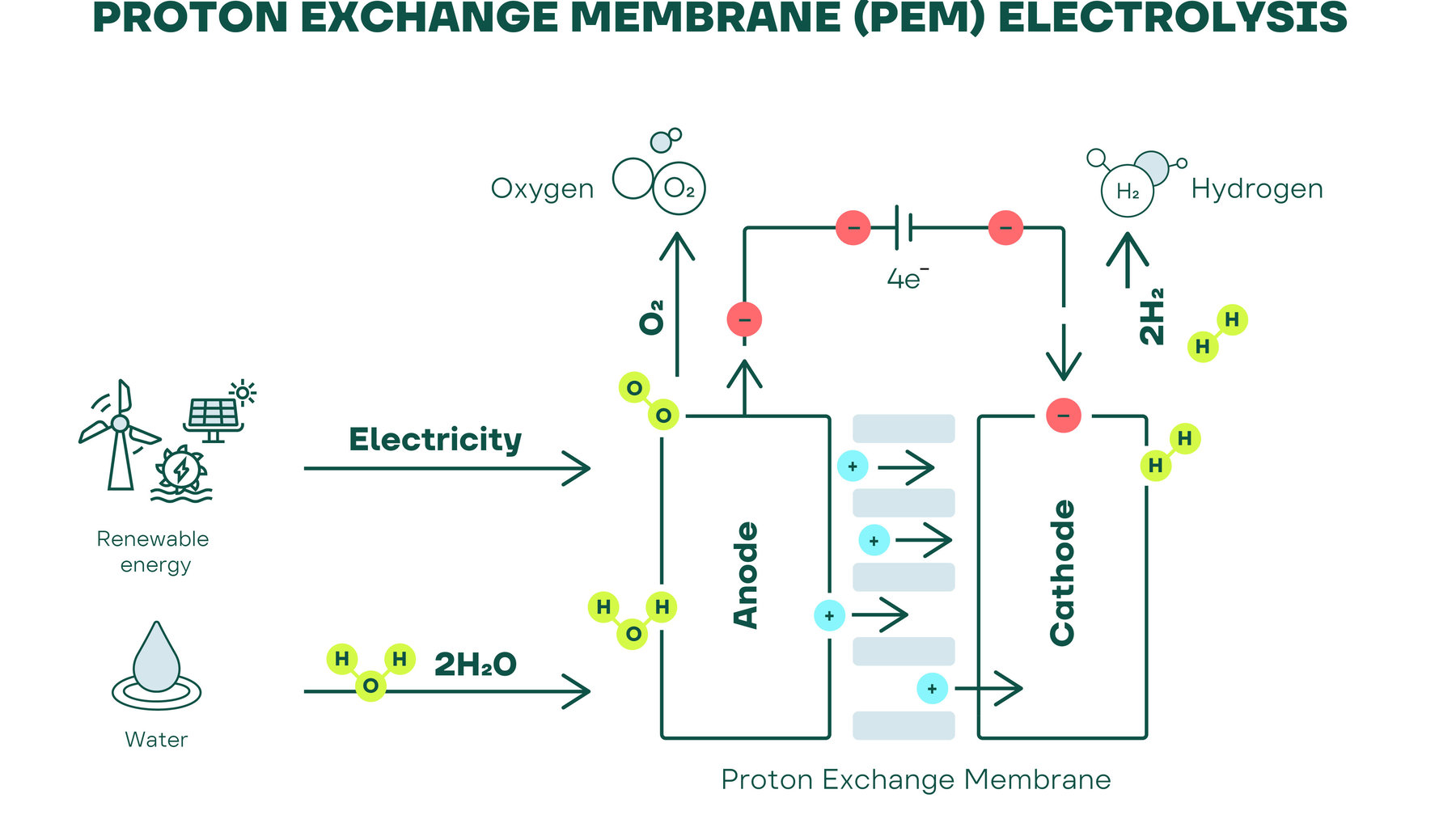
Hydrogen production by electrolysis was described as early as 1800 and is one of the oldest electrochemical processes. Hydrogen electrolysis uses electricity to split water into hydrogen and oxygen: Two water molecules (2 H2O) each become two hydrogen molecules (2 H2) and one oxygen molecule (O2).
Electrolysis requires two electrodes, an anode and a cathode, direct current and a liquid electrolyte for alkaline electrolysis or an acidic solid electrolyte membrane for PEM electrolysis. The use of direct current separates the water into its components. The hydrogen collects at the negatively charged cathode and the oxygen at the positively charged anode.
Alkaline electrolysis
Alkaline electrolysis is almost 100 years old and is the oldest technology available on a large scale. Alkaline electrolysis uses potassium hydroxide (KOH) as the electrolyte. There is a diaphragm between the cathode and anode that is impermeable to the product gases hydrogen and oxygen, preventing them from mixing. It also prevents the formation of explosive gases. The negatively charged hydroxide ions can pass through the diaphragm and move to the anode. The potassium hydroxide solution is constantly circulated, ensuring continuous degassing.
However, alkaline electrolyzers can be less dynamic in their response to load changes. For this reason, alkaline electrolysis has some disadvantages when coupled with renewable electricity sources: For example, the achievable gas purity decreases in partial load operation and degradation problems occur. One reason for this is the relatively sluggish electrolyte cycle. In addition, alkaline electrolysis requires a longer cold start time.
PEM electrolysis
PEM electrolysis was developed about 50 years ago. This makes it the newer technology, but like alkaline electrolysis, it is already commercially available on an industrial scale. Instead of a liquid electrolyte, PEM electrolysis uses a solid polymer electrolyte, the proton-conducting “proton exchange membrane” or “polymer electrolyte membrane” (PEM).
PEM electrolysis is essentially the reverse of the fuel cell principle. A PEM electrolyzer is used to convert water into hydrogen and oxygen. The process takes place in the electrolysis stack, the heart of the electrolyzer. During electrolysis, positively charged hydrogen ions (protons) move through the gas-tight membrane to the cathode. There they combine with an electron to form hydrogen molecules. At the same time, oxygen is separated on the anode side, which can be reused depending on the system technology. The gas-impermeable membrane ensures that the electrolysis products hydrogen and oxygen do not mix. Unlike alkaline electrolysis, the PEM electrolyzer requires only high-purity, deionized water in the internal water circuit. The use of this water helps to protect the catalysts from impurities, preventing degradation and the gradual loss of performance and efficiency.
The use of highly purified, deionized water also helps to ensure that the hydrogen produced is of a higher purity than other electrolysis technologies.